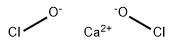Calcium hypochlorite CAS#7778-54-3
CAS Number: 7778-54-3
Chemical Formula: C38H60O18
Synonyms:
Calcium hypochlorite technical grade
calciumb11306hypochlorite
calciumchlorohydrochlorite
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: White Powder
Calcium hypochlorite CAS#7778-54-3
Calcium hypochlorite (molecular formula: Ca (ClO)2) is a kind of inorganic compound. It appears as a white granular solid with chlorine odor. Though it is relatively stable and non-combustible, it will accelerate the burning of combustible materials. Sodium hypochlorite solid is generally not commercialized. Instead, it is highly soluble in water and can be formulated into various concentrations. The resulting sodium hypochlorite solutions appears as clear, green to yellow liquids. It has two major applications. One is sanitation. For this purpose, it is primarily used as bleaching agent or disinfectants to sanitize publish swimming pools as well as disinfect drinking water. It is also used in the disinfection of surfaces and equipment of kitchen and bathroom. Moreover, it can be used as algaecides, herbicide and laundry detergents. Another major application is in organic chemistry. It can act as a general oxidizing agent for the cleavage of glycols, keto acids to obtain fragmented aldehydes or carboxylic acid. It can also be used for the manufacture of chloroform. Calcium hypochlorite can be manufactured through the reaction between limes (Ca(OH)2) with chlorine gas to give various concentrations of products. Calcium hypochlorite is toxic. Its solution can emit toxic gases such as chlorine. Inhalation can lead to olfactory fatigue and irritation on the lung airway. It is also irritating to the skin and eye upon contact. Ingestion of it can cause vomiting and corrosive injury to the gastrointestinal tract.
Calcium hypochlorite Chemical Properties |
Melting point | 100 °C(lit.) |
density | 2.35 g/mL at 25 °C(lit.) |
refractive index | 1.545 |
storage temp. | Store at +15°C to +25°C. |
solubility | 200g/l (decomposition) |
form | Tablets |
color | White to off-white or faint green |
Specific Gravity | 2.35 |
Odor | Chlorine-like odor |
Water Solubility | 200 g/L (20 ºC) (dec.) |
Merck | 14,1674 |
Dielectric constant | 2.3(Ambient) |
Stability: | Stability Strong oxidizer - contact with flammable material may lead to fire. Incompatible with water, reducing agents, combustible material, phenol. |
Cosmetics Ingredients Functions | ANTIMICROBIAL |
InChI | 1S/Ca.2ClO/c;2*1-2/q+2;2*-1 |
InChIKey | ZKQDCIXGCQPQNV-UHFFFAOYSA-N |
SMILES | ClO[Ca]OCl |
CAS DataBase Reference | 7778-54-3(CAS DataBase Reference) |
EPA Substance Registry System | Calcium hypochlorite (7778-54-3) |
Safety Information |
Hazard Codes | O,C,N |
Risk Statements | 8-22-31-34-50 |
Safety Statements | 26-36/37/39-45-61 |
RIDADR | UN 1748 5.1/PG 2 |
WGK Germany | 2 |
RTECS | NH3485000 |
TSCA | TSCA listed |
HS Code | 2828 10 00 |
HazardClass | 5.1 |
PackingGroup | II |
Storage Class | 5.1A - Strongly oxidizing hazardous materials |
Hazard Classifications | Acute Tox. 4 Oral |
Hazardous Substances Data | 7778-54-3(Hazardous Substances Data) |
Toxicity | LD50 orally in Rabbit: 850 mg/kg LD50 dermal Rabbit > 2000 mg/kg |
Product Application Of Calcium hypochlorite CAS#7778-54-3
Deodorant, oxidizing agent, bleaching agentCalcium hypochlorite is used for swimming pool sanitation and other water treatment plants. It is also used to disinfect drinking water, equipments in the industry and surfaces and equipments in kitchens. Further, it is used to bleach textiles and paper. It is also employed in bathroom cleaners and laundry detergents. It is used as an oxidizing agent in organic chemistry. It is involved in the cleavage of glycols, alfa-hydroxy carboxylic acids and keto acids to yield fragmented aldehydes or carboxylic acids respectively. It plays an important role in the haloform reaction to prepare chloroform.
Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days