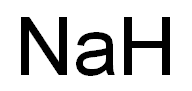Sodium hydride CAS#7646-69-7
CAS Number: 7646-69-7
Chemical Formula: HNa
Synonyms:
SODIUM HYDRIDE
SODIUM HYDRIDE DISPERSION
NAH 80;0.84-0.86
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: White Powder
Sodium hydride CAS#7646-69-7
Sodium hydride, is a binary salt that has a specific hazard of releasing hydrogen upon contact with water. It is an odorless powder that is violently water reactive. The four-digit UN identification number is 1427. The NFPA 704 designation is health 3, flammability 3, and reactivity 2. The white space at the bottom of the diamond has a W with a slash through it, indicating water reactivity. |
Sodium hydride Chemical Properties |
Melting point | 800 °C (dec.) (lit.) |
density | 1.2 |
Fp | 185°C |
storage temp. | Store below +30°C. |
solubility | Soluble in molten sodium. Insoluble in ammonia, benzene,carbon tetrachloride, carbon disulfide and all organic solvents. |
form | powder (moistened with oil) |
color | White to pale gray |
Water Solubility | REACTS |
Sensitive | Air & Moisture Sensitive |
Merck | 14,8625 |
Exposure limits | ACGIH: TWA 5 mg/m3 |
InChI | InChI=1S/Na.H |
InChIKey | MPMYQQHEHYDOCL-UHFFFAOYSA-N |
SMILES | [NaH] |
CAS DataBase Reference | 7646-69-7(CAS DataBase Reference) |
NIST Chemistry Reference | Sodium hydride(7646-69-7) |
EPA Substance Registry System | Sodium hydride (NaH) (7646-69-7) |
Safety Information |
Hazard Codes | F,C |
Risk Statements | 15-36-35-11 |
Safety Statements | 26-43-7/8-43A-24/25-7-60-22-45-37/39-16 |
RIDADR | UN 1427 4.3/PG 1 |
WGK Germany | 3 |
RTECS | WB3910000 |
F | 10-21 |
TSCA | TSCA listed |
HazardClass | 4.3 |
PackingGroup | I |
HS Code | 28500090 |
Storage Class | 4.3 - Hazardous materials which set free flammable gases upon contact with water |
Hazard Classifications | Eye Dam. 1 |
Hazardous Substances Data | 7646-69-7(Hazardous Substances Data) |
Product Application Of Sodium hydride CAS#7646-69-7
Sodium hydride is used to enhance the condensation reactions of carbonyl compounds through the Dieckmann condensation, Stobbe condensation, Darzens condensation and Claisen condensation. It acts as a reducing agent used to prepare diborane from boron trifluoride. It is also used in fuel cell vehicles. Further, it is used to dry some organic solvents. In addition to this, it is involved in the preparation of sulfur ylides, which is utilized for the conversion of ketones into epoxides.
Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days