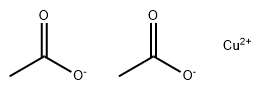
Copper(II) acetate CAS#142-71-2
Copper (II) acetate, also referred to as cupric acetate, is the chemical compound with the formula Cu(OAc)22 where OAc- is acetate (CH3CO2-). The hydrated derivative, which contains one molecule of water for each Cu atom, is available commercially. Anhydrous Cu(OAc)2 is a dark green crystalline solid, whereas Cu2(OAc)4(H2O)2 is more bluish-green. Since ancient times, copper acetates of some form have been used as fungicides and green pigments. Today, copper acetates are used as reagents for the synthesis of various inorganic and organic compounds. Copper acetate, like all copper compounds, emits a blue-green glow in a flame.
Copper(II) acetate Chemical Properties |
Melting point | 115°C |
density | 1.92 at 21.9℃ |
vapor density | 6.9 (vs air) |
vapor pressure | 0.002Pa at 25℃ |
storage temp. | Inert atmosphere,Room Temperature |
solubility | |
form | Powder |
color | Green to blue |
Flame Color | Green |
Water Solubility | Soluble in water and alcohol. Slightly soluble in ether and glycerol. |
Sensitive | Hygroscopic |
Merck | 14,2624 |
BRN | 3595638 |
Exposure limits | ACGIH: TWA 1 mg/m3 |
Stability: | hygroscopic |
Cosmetics Ingredients Functions | SKIN CONDITIONING |
InChI | 1S/2C2H4O2.Cu/c2*1-2(3)4;/h2*1H3,(H,3,4);/q;;+2/p-2 |
InChIKey | OPQARKPSCNTWTJ-UHFFFAOYSA-L |
SMILES | CC(=O)O[Cu]OC(C)=O |
LogP | -0.285 (est) |
Surface tension | 72mN/m at 1.01-1.08g/L and 21.2-21.6℃ |
Dissociation constant | 4.69-4.89 |
CAS DataBase Reference | 142-71-2(CAS DataBase Reference) |
EPA Substance Registry System | Cupric acetate (142-71-2) |
Safety Information |
Hazard Codes | Xn,N |
Risk Statements | 22-36/37/38-50/53 |
Safety Statements | 26-60-61-37/39-29 |
RIDADR | UN 3077 9/PG 3 |
WGK Germany | 3 |
RTECS | AG3480000 |
F | 3-10 |
TSCA | TSCA listed |
HazardClass | 9 |
HS Code | 29152990 |
Storage Class | 8B - Non-combustible corrosive hazardous materials |
Hazard Classifications | Acute Tox. 4 Oral |
Hazardous Substances Data | 142-71-2(Hazardous Substances Data) |
Toxicity | mouse,LD50,intraperitoneal,2500ug/kg (2.5mg/kg),Biochemical Pharmacology. Vol. 30, Pg. 771, 1981. |
Product Application Of Copper(II) acetate CAS#142-71-2
The uses for copper (II) acetate are more plentiful as a catalyst or oxidizing agent in organic syntheses. For example, Cu2(OAc)4 is used to couple two terminal alkynes to make a 1,3-diyne:
Cu2(OAc)4 + 2 RC ≡ CH → 2 CuOAc + RC ≡ C-C ≡ CR + 2 HOAc
The reaction proceeds via the intermediacy of copper(I) acetylides, which are then oxidized by the copper(II) acetate, releasing the acetylide radical. A related reaction involving copper acetylides is the synthesis of ynamines, terminal alkynes with amine groups using Cu2(OAc)4.
Fact Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days