Bromoethane CAS#74-96-4
CAS Number: 74-96-4
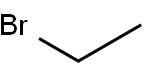
Chemical Formula: C2H5Br
Synonyms:
Bromoethane, extra pure
1-Bromethan;Bromethan
ETHYL BROMIDE extrapure
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: Transperant Liquid
Bromoethane CAS#74-96-4
Bromoethane is a colorless, volatile, flammable liquid. When exposed to air and light, it turns yellow. It has an ethereal odor and somewhat burning taste. Bromoethane has a specific gravity of 1.4505 between 4° and 25°C, a boiling point of 38.4°C, a melting point of -119°C, and a vapor pressure of 475 mm mercury at 25°C. It is 0.91% (w/w) soluble in water at 20°C and is miscible with ethanol, ethyl ether, chloroform, and other organic solvents. It has a flash point of -20°C (closed cup). The autoignition temperature is 511°C. The flammable limits in air are between 6.7570 and 11.25%. Although bromoethane is relatively stable, when heated to decomposition it emits highly toxic fumes of bromine and hydrobromide; it can react with oxidizing materials (ITII, 1979; Sittig, 1979; Torkelson and Rowe, 1981; Merck, 1983; Sax, 1984).
Bromoethane Chemical Properties |
Melting point | -119 °C |
Boiling point | 37-40 °C(lit.) |
density | 1.46 g/mL at 25 °C(lit.) |
vapor density | ~3.75 (vs air) |
vapor pressure | 25.32 psi ( 55 °C) |
refractive index | n |
Fp | -23 °C |
storage temp. | Store below +30°C. |
solubility | Miscible with ethanol, ether, chloroform and organic solvents. |
form | Liquid |
color | Clear colorless to slightly yellow |
explosive limit | 6.7-11.3%(V) |
Water Solubility | 0.91 g/100 mL (20 ºC) |
Sensitive | Light Sensitive |
Merck | 14,3771 |
BRN | 1209224 |
Henry's Law Constant | 7.56(x 10-3 atmm3/mol) at 25 °C (Hine and Mookerjee, 1975) |
Dielectric constant | 9.4(20℃) |
Exposure limits | TLV-TWA 200 ppm (~90 mg/m3) (ACGIH, MSHA, OSHA, and NIOSH); TLV STEL 250 ppm (~110 mg/m3) (ACGIH); IDLH 3500 ppm (NIOSH). |
Stability: | Stable. Highly flammable. Readily forms explosive mixtures with air. Note low flash point. Incompatible with alkali metals, aluminium, magnesium, strong bases, water, strong oxidizing agents. May be light sensitive. |
InChI | 1S/C2H5Br/c1-2-3/h2H2,1H3 |
InChIKey | RDHPKYGYEGBMSE-UHFFFAOYSA-N |
SMILES | CCBr |
Surface tension | 24.2mN/m at 20°C |
CAS DataBase Reference | 74-96-4(CAS DataBase Reference) |
NIST Chemistry Reference | Ethane, bromo-(74-96-4) |
IARC | 3 (Vol. 52, 71) 1999 |
EPA Substance Registry System | Bromoethane (74-96-4) |
Safety Information |
Hazard Codes | F,Xn |
Risk Statements | 11-20/22-40 |
Safety Statements | 36/37 |
RIDADR | UN 1891 6.1/PG 2 |
WGK Germany | 1 |
RTECS | KH6475000 |
F | 8 |
Autoignition Temperature | 952 °F |
TSCA | TSCA listed |
HS Code | 2903 39 19 |
HazardClass | 6.1 |
PackingGroup | II |
Storage Class | 3 - Flammable liquids |
Hazard Classifications | Acute Tox. 4 Inhalation |
Hazardous Substances Data | 74-96-4(Hazardous Substances Data) |
Toxicity | LC50 rats, mice (ppm): 27000, 16200 (Vernot) |
IDLA | 2,000 ppm |
Product Application of Bromoethane CAS#74-96-4
Bromoethane is an alkylating agent primarily used as a chemical intermediate in organic synthesis, in the manufacture of pharmaceuticals, and for the ethylation of gasoline. To a lesser extent, it has been used as a fruit and grain fumigant, refrigerant, and solvent. Although proposed occasionally as a general anesthetic in the earlier part of this century, it has not been used to any extent for this purpose (Sayers et al., 1929; Abreu et al., 1939; ITII, 1979; Sittig, 1979; Torkelson and Rowe, 1981;Merck, 1983).
Fact Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days