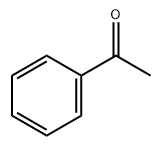
Acetophenone CAS#98-86-2
CAS Number: 98-86-2
Chemical Formula: C8H8O
Synonyms:
ACETOPHENONE, 1X1ML, CH2CL2, 2000UG/ML
ACETOPHENONE extrapure AR
METHYL PHENYL KETONE
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: Transperant Liquid
Acetophenone CAS#98-86-2
Acetophenone is also known as acetyl benzene with the boiling point (℃) being 202.3, relative density (water = 1) being 1.03 (20 ℃) and the relative vapor density (air = 1) being 4.14. It is the simplest aromatic ketones with its aromatic core (benzene ring) being directly connected with a carbonyl group. It is presented in some kinds of essential oils of some plants in its free-state form. It is pure colorless crystals. Most commercially available product appears as pale yellow oily liquid with hawthorn like aroma. It is only slightly soluble in water and is easily soluble in many organic solvents and can be evaporated together with the steam. The molecular structure of acetophenone: methyl C atom is bonded through sp3 hybrid orbital while the benzene ring and the carbonyl C atom are bonded through sp2 hybrid orbital. Addition reaction and [alpha] active hydrogen reaction can occur for the carbonyl group of acetophenone. It can also have electrophilic substitution reaction in its benzene ring with the major product generated being in the meta-position. Acetophenone can be produced from the reaction between benzene with acetyl chloride, acetic anhydride or acetate under the catalysis of aluminum trichloride. Furthermore, when ethylbenzene is catalyzed and oxidized into styrene, it can also generate acetophenone as by-product. Acetophenone is mainly used as the raw materials for pharmaceuticals and other kind of organic synthesis. It can also used for the preparation of spices, soap and cigarettes as well as being used as the solvents of cellulose ether, cellulose ester and resin and plasticizer. It has hypnotic effect. Currently, acetophenone is mostly obtained via the by-product of reaction between phenol and acetone via cumene oxidation. It may be obtained through the acetylation on benzene through acetyl chloride.
Acetophenone Chemical Properties |
Melting point | 19-20 °C (lit.) |
Boiling point | 202 °C (lit.) |
density | 1.03 g/mL at 25 °C (lit.) |
vapor density | 4.1 (vs air) |
vapor pressure | 0.45 mm Hg ( 25 °C) |
refractive index | n |
FEMA | 2009 | ACETOPHENONE |
Fp | 180 °F |
storage temp. | Store below +30°C. |
solubility | 6.1g/l |
form | Liquid |
color | Clear colorless to light yellow |
Relative polarity | 4.4 |
Odor | Pungent, floral odor |
Odor Type | floral |
explosive limit | 1.4-5.2%(V) |
biological source | synthetic |
Water Solubility | 5.5 g/L (20 ºC) |
Merck | 14,73 |
JECFA Number | 806 |
BRN | 605842 |
Exposure limits | No exposure limits are set. The health hazard from exposure to this compound should be low, due to its low vapor pressure and low toxicity. |
Dielectric constant | 17.4(25℃) |
Stability: | Stable. Incompatible with strong oxidizing agents, strong bases, strong reducing agents. Combustible. |
Major Application | flavors and fragrances |
Cosmetics Ingredients Functions | PERFUMING |
InChI | 1S/C8H8O/c1-7(9)8-5-3-2-4-6-8/h2-6H,1H3 |
InChIKey | KWOLFJPFCHCOCG-UHFFFAOYSA-N |
SMILES | CC(=O)c1ccccc1 |
LogP | 1.65 at 20℃ |
Surface tension | 39.54 mN/m at 283.15K |
CAS DataBase Reference | 98-86-2(CAS DataBase Reference) |
NIST Chemistry Reference | Acetophenone(98-86-2) |
EPA Substance Registry System | Acetophenone (98-86-2) |
Safety Information |
Hazard Codes | Xn,T,F |
Risk Statements | 22-36-63-43-36/37/38-23/24/25-45-39/23/24/25-11-67-40 |
Safety Statements | 26-36/37-24/25-23-53-45-16-7 |
RIDADR | UN 1593 6.1/PG 3 |
WGK Germany | 1 |
RTECS | AM5250000 |
F | 8 |
Autoignition Temperature | 570 °C |
TSCA | TSCA listed |
HazardClass | 9 |
PackingGroup | III |
HS Code | 29143900 |
Storage Class | 10 - Combustible liquids |
Hazard Classifications | Acute Tox. 4 Oral |
Hazardous Substances Data | 98-86-2(Hazardous Substances Data) |
Toxicity | LD50 orally in rats: 0.90 g/kg (Smyth, Carpenter) |
Product Application of Acetophenone CAS#98-86-2
Methyl phenyl ketone is mainly converted to resins (synthetic resins) by reaction with formaldehyde. It is also used as a photoinitiator for special printing plates as well as for organic syntheses, particularly for pharmaceuticals. Certain derivatives, such as 1-phenylethanol (obtained by hydrogenation) and its acetate, are used as fragrances. Furthermore, methyl phenyl ketone is used for the synthesis of optical brighteners.
Fact Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days