1,2-Dichloroethane CAS#107-06-2
CAS Number: 107-06-2
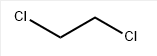
Chemical Formula: C2H4Cl2
Synonyms:
1,2-dichloroethane (ethylene dichloride)
1,2-Bichloroethane
1,2-Dichlor-aethan
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: Transperant Liquid
1,2-Dichloroethane CAS#107-06-2
1,2-Dichloroethane, also called ethylene dichloride (EDC), is a manufactured chemical that is not found naturally in the environment. It is used principally to synthesize vinyl chloride, which is further used to produce a variety of vinyl based plastics products, such as polyvinyl chloride (PVC) pipes, furniture, automobile upholstery, wall coverings, housewares, and automobile parts. It is used in solvents in closed systems for various extraction and cleaning purposes in organic synthesis. It is used as a leaded gasoline additive to remove lead, but with declining tendency. It is used as a dispersant in rubber and plastics, as a wetting and penetrating agent. It was used in ore flotation, as a metal degreaser, and in textile and PVC cleaning. It was also used as an insect fumigant for stored grains and in mushroom houses, a soil fumigant in peach and apple orchards. But due to its toxicity, it is no longer registered for use as an insect fumigant in the United States (IARC 1999).
1,2-Dichloroethane Chemical Properties |
Melting point | -35 °C (lit.) |
Boiling point | 83 °C (lit.) |
density | 1.256 g/mL at 25 °C (lit.) |
vapor density | 3.4 (20 °C, vs air) |
vapor pressure | 87 mm Hg ( 25 °C) |
refractive index | n |
Fp | 60 °F |
storage temp. | 0-6°C |
solubility | 7.9g/l |
form | Liquid |
color | APHA: ≤10 |
Odor | Chloroform-like odor |
Relative polarity | 0.327 |
explosive limit | 6.2-15.9%(V) |
Water Solubility | 8.7 g/L (20 ºC) |
Merck | 14,3797 |
BRN | 605264 |
Henry's Law Constant | 11.24 at 30 °C (headspace-GC, Sanz et al., 1997) |
Exposure limits | TLV-TWA 10 ppm (~40 mg/m3) (ACGIH), 1 ppm (NIOSH), 50 ppm (MSHA and OSHA); ceiling 2 ppm/15 min (NIOSH); carcinogenicity: Animal Sufficient Evidence, Human Limited Evidence (IARC). |
Dielectric constant | 10.7(20℃) |
Stability: | Volatile |
Cosmetics Ingredients Functions | NOT REPORTED |
InChI | 1S/C2H4Cl2/c3-1-2-4/h1-2H2 |
InChIKey | WSLDOOZREJYCGB-UHFFFAOYSA-N |
SMILES | ClCCCl |
LogP | 1.45 at 20℃ |
Surface tension | 33.3mN/m at 20°C |
CAS DataBase Reference | 107-06-2(CAS DataBase Reference) |
IARC | 2B (Vol. 20, Sup 7, 71) 1999 |
NIST Chemistry Reference | Ethane, 1,2-dichloro-(107-06-2) |
EPA Substance Registry System | 1,2-Dichloroethane (107-06-2) |
Safety Information |
Hazard Codes | F,T |
Risk Statements | 45-11-22-36/37/38-23/25-23 |
Safety Statements | 53-45-24-16-7 |
RIDADR | UN 1184 3/PG 2 |
OEB | B |
OEL | TWA: 1 ppm (4 mg/m3), STEL: 2 ppm (8 mg/m3) (Chloroethanes) |
WGK Germany | 3 |
RTECS | KI0525000 |
F | 3-10 |
Autoignition Temperature | 775 °F |
TSCA | TSCA listed |
HS Code | 2903 15 00 |
HazardClass | 3 |
PackingGroup | II |
Storage Class | 3 - Flammable liquids |
Hazard Classifications | Acute Tox. 3 Inhalation |
Hazardous Substances Data | 107-06-2(Hazardous Substances Data) |
Toxicity | LD50 orally in rats: 770 mg/kg (Smyth) |
IDLA | 50 ppm |
Product Application of 1,2-Dichloroethane CAS#107-06-2
1,2-dichloroethane (Ethylene dichloride), also known as EDC, is produced by reacting chlorine or anhydrous hydrochloric acid with ethylene. The largest single use for EDC is the production of vinyl chloride monomer, which is used to produce poly vinyl chloride (PVC). It has many uses in industry, with principal ones being the following:
As an intermediate in the manufacture of methyl chloroform, perchloroethylene, ethylene amines, polyvinyl chloride (PVC), sulfide compounds, vinyl chloride, and trichloroethane.
As an additive in gasoline (used as a lead scavenger), pharmaceutical products, color film, and pesticides.
Fact Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days