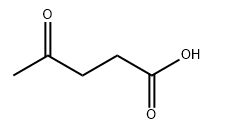
Levulinic acid CAS#123-76-2
CAS Number: 123-76-2
Chemical Formula: C5H8O3
Synonyms:
sodium 4-oxopentanoate
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: Transperant Yellow Liquid
Levulinic acid has a tart, whiskey taste. It may be synthesized by the action of more or less concentrated HCL on sucrose, glucose, or fructose; hence, its reported presence in caramels.4-oxopentanoic acid is an oxopentanoic acid with the oxo group in the 4-position. It has a role as a plant metabolite. It is a straight-chain saturated fatty acid and an oxopentanoic acid. It is a conjugate acid of a 4-oxopentanoate.
Levulinic acid Chemical Properties |
Melting point | 30-33 °C (lit.) |
Boiling point | 245-246 °C (lit.) |
density | 1.134 g/mL at 25 °C (lit.) |
vapor pressure | 1 mm Hg ( 102 °C) |
refractive index | n |
FEMA | 2627 | LEVULINIC ACID |
Fp | 280 °F |
storage temp. | Store below +30°C. |
solubility | 675g/l |
form | Liquid After Melting |
pka | pKa 4.65(H2O,t = 25,c=0.03-0.001) (Uncertain) |
color | Clear yellow |
Odor | at 100.00 %. sweet caramel acidic acetoin buttery |
Odor Type | caramellic |
biological source | synthetic |
Water Solubility | Soluble in water ( 675g/L at 20°C). |
Sensitive | Light Sensitive |
Merck | 14,5472 |
JECFA Number | 606 |
BRN | 506796 |
Cosmetics Ingredients Functions | PERFUMING |
InChIKey | JOOXCMJARBKPKM-UHFFFAOYSA-N |
LogP | -0.498 |
Surface tension | 42.53mN/m at 298.15K |
CAS DataBase Reference | 123-76-2(CAS DataBase Reference) |
NIST Chemistry Reference | Pentanoic acid, 4-oxo-(123-76-2) |
EPA Substance Registry System | Levulinic acid (123-76-2) |
Safety Information |
Hazard Codes | Xn,C |
Risk Statements | 22-36/37/38-34-R34-R22 |
Safety Statements | 26-45-36/37/39-S45-S36/37/39-S26 |
RIDADR | 3261 |
WGK Germany | 3 |
RTECS | OI1575000 |
TSCA | TSCA listed |
HazardClass | 8 |
PackingGroup | III |
HS Code | 29183000 |
Toxicity | LD50 orally in Rabbit: 1850 mg/kg LD50 dermal Rabbit > 5000 mg/kg |
Product Application of Levulinic acid CAS#123-76-2
Levulinic acid is a precursor for the synthesis of useful intermediates such as γ-valerolactone, ethyl levulinate, pentanoic acid and 2-methyl-tetrahydrofuran. Derivatization and esterification of levulinic acid results in potential biofuels.
It can also be used in:
The preparation of catalytic composite to synthesize 5-hydroxymethylfurfural and furfural.
The synthesis of a commercial fragrance, fraistone.
The synthesis of pyrrolidone derivatives via reductive amination.
The total synthesis of mycobacterial arabinogalactan.
Factory and Equipment Show
Fast delivery time
Inventory 2-3 working days New production 7-10 working days